Retractions are a serious threat to biomedical research
In the high-stakes world of biomedical research, where published findings can shape clinical practice, policy decisions, and even drug approvals, the presence of retracted literature is not just an academic problem, it’s a public health concern. When flawed, fabricated, or irreproducible studies are left unchecked in the scientific ecosystem, they continue to misinform downstream research, meta-analyses, clinical guidelines, and ultimately, patient care.
Retractions aren’t rare, either. According to Retraction Watch, retractions have been steadily rising over the last decade. Still, many retracted studies continue to circulate in the literature without any obvious indication that they’ve been pulled.
AI-powered biomedical searching and retractions
There are dozens, maybe even hundreds, of AI tools that promise to revolutionize biomedical literature searching. These tools claim to make life easier for clinicians and researchers by surfacing “the best” evidence quickly.
Unfortunately, these AI tools likely struggle with reliably flagging retracted articles. None of these tools appear to cross-reference the Retraction Watch Database, even though it’s one of the most comprehensive and up-to-date sources of retraction data.
The result? Users could end up citing, summarizing, or even basing treatment decisions on debunked science and the AI tools they trusted helped them do it.
Putting three AI search assistant tools to the test
To assess whether current AI-powered tools can reliably detect and communicate retracted biomedical research, we ran a small, but telling test using a recently retracted article:
Wu, S. Y., Sharma, S., Wu, K., Tyagi, A., Zhao, D., Deshpande, R. P., & Watabe, K. (2021). Tamoxifen suppresses brain metastasis of estrogen receptor-deficient breast cancer by skewing microglia polarization and enhancing their immune functions. Breast Cancer Research, 23, 1-16.
This article was retracted on May 12, 2025.
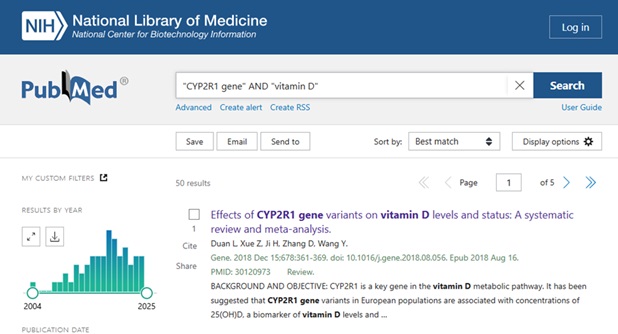
We located this article through the Retraction Watch Database, a critical resource for identifying retracted papers. We then tested how three popular AI tools responded when we searched for this article: 1) SciSpace, 2) Consensus, and 3) Elicit/
Baseline: Publisher and PubMed got it right
The article is clearly marked as retracted on both the publisher’s website (BMC, part of Springer Nature) and in PubMed. On BMC’s site, the article is branded with a bold red banner indicating that it has been retracted, and it links directly to the retraction notice.
In PubMed, the article’s retraction status is labelled. There’s a large red “Retracted Article” warning at the top of the article record.

With Third Iron’s LibKey Nomad browser extension installed, the retraction warning also appeared directly in the search results list, providing an extra layer of protection.

These platforms demonstrate that it is possible to handle retractions clearly and transparently. But what happens when you try to search with an AI-powered tool?
1) SciSpace: No retraction flag, no awareness
SciSpace has gained traction for its AI-enabled “Papers” database and its Chat AI for article summarization. We searched for the retracted article using the Papers function. The article was retrieved with no indication that it had been retracted.
The PDF version offered by SciSpace appeared to be the original, unretracted version of the paper — there was no watermark or retraction notice. This likely occurred because SciSpace stored an earlier version of the file and does not dynamically update with retraction metadata or new PDFs.
When we asked the SciSpace Chat if the article had been retracted, the reply was: “Sorry, this is not discussed in the paper.” In other words, the AI agent only read the text of the article and had no external awareness of its retraction status.

SciSpace also failed to locate or return the associated Retraction Note (PMID: 40355962), which was published in the same journal.
2) Consensus: Accurate link, but no warning
Consensus is designed to help users quickly identify answers to scientific questions by ranking statements from published articles.
The article was returned in a basic search, and no indication of its retracted status was provided. The PDF link routed to the publisher’s version, which is good practice. Since BMC properly flags retractions, users landing on that page would see the retraction banner and be able to access the Retraction Note. While Consensus did not flag the article as retracted in its own search interface or metadata, it did link out to a source that did.

3) Elicit: Somewhat better!
Elicit offers two formats for reviewing articles: a plain-text view and a PDF view. When we searched for the retracted article via Elicit’s “Find Papers” tool, the results were mixed.

The article summary did not indicate that the paper had been retracted. The plain text view contained the word “RETRACTED” throughout the body text. Elicit also linked to a newer version of the article PDF that had the retraction stamp clearly watermarked across every page.

Lessons learned: We need accountability and standards
Users skimming article summaries, relying on search results, or using data extraction tables generated by AI tools might still miss the retraction unless they click deeper into the article itself. This is especially concerning in evidence synthesis workflows, where tools like Elicit auto-populate summary tables with study characteristics and conclusions—often without indicating the article has been retracted.
If AI is going to play a meaningful role in evidence retrieval and synthesis, it needs to be held to a higher standard. At a minimum, AI tools used in biomedical contexts must:
- Flag retracted articles clearly and automatically
- Cross-reference multiple retraction sources, including Retraction Watch
- Date-stamp and cite their information sources transparently
- Allow users to report errors or omissions easily
Until the current AI tools ecosystem improves, here are some tips to protect yourself and your team:
- Always cross-check critical articles in the Retraction Watch Database or in PubMed
- Use reference managers (like Zotero or EndNote) that integrate with PubMed and allow for manual annotations of retracted status
- Avoid relying solely on AI summaries or ranking algorithms, especially for high-stakes research
It’s also worth noting that all the tools we tested (SciSpace, Consensus, and Elicit) are paid products and are effectively marketed as intelligent research assistants. Yet their inconsistent handling of retracted literature highlights the need for human-level vetting and cross-referencing.
In practice, this can take significantly more time than a traditional search if you’re trying to be thorough. Instead of accelerating research, these tools often introduce a false sense of efficiency, making it easy to miss red flags that would be obvious in a well-curated, librarian-led search process. The MSK Library team can help you navigate retraction risks, validate sources, and choose the right tools for your research. Connect with us today.